Personalised risk stratification in prostate cancer and digital pathology
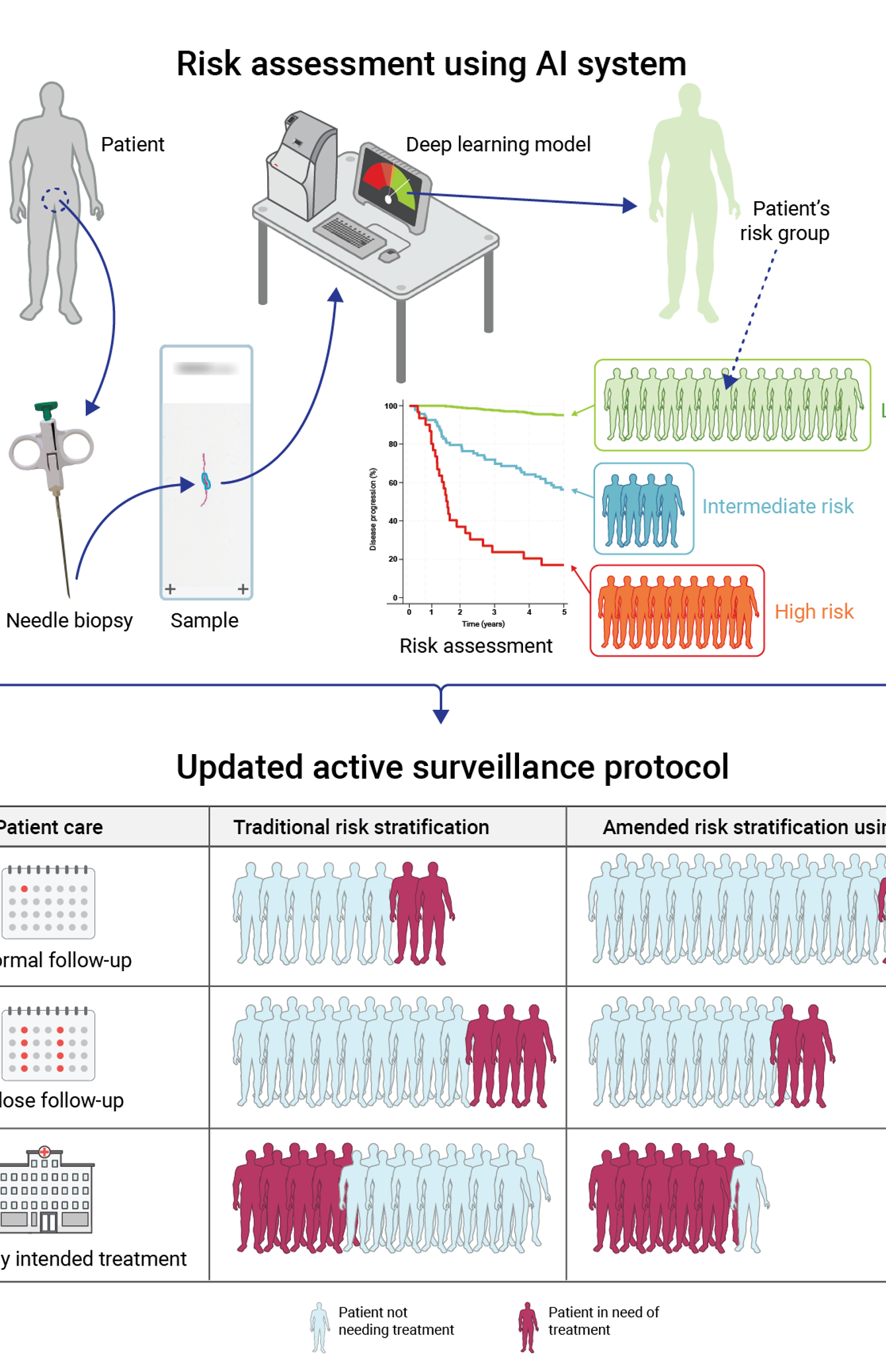
One of the greatest challenges in prostate cancer care is identifying which patients need immediate treatment and which can be safely monitored. In this project, digital pathology and artificial intelligence (AI) are used to develop a more precise tool for risk assessment at the time of diagnosis.
Continuing the development of a deep learning model originally optimised for surgical specimens, the project will adapt this approach to diagnostic tissue samples (biopsies) to better assess tumour aggressiveness at the time of initial disease detection. The aim is to reduce unnecessary overtreatment by improving selection for active surveillance, while ensuring timely treatment for patients with aggressive disease.
The dilemma in current prostate cancer care
Prostate cancer is a global health challenge and the most common cancer in men in Norway, with over 5,200 new diagnoses annually. While PSA testing and MRI-guided biopsies have improved early detection of prostate cancer, they have also led to a major clinical dilemma: balancing the benefits of early diagnosis against the risk of overtreatment.
For patients diagnosed with localised cancer, the choice is between potentially curative but invasive treatments (such as removal of the prostate or radiotherapy) and “active surveillance”. Active surveillance means that the patient is monitored with frequent check-ups, and treatment is only initiated if there are indications of cancer progression. The challenge, however, is that today’s standard tools for assessing risk are imprecise. As a result, about half of these patients end up in a vague “intermediate risk” group, where the choice between surveillance and treatment is uncertain, leading to substantial variation in management between hospitals.
The gap between research and clinical practice
To make safer decisions about who should undergo surgery and who can be monitored, better prognostic tools are needed at the time of diagnosis. In current cancer research, new biomarkers are often developed by studying large amounts of tissue from entire prostate glands that have been surgically removed. But when the actual treatment decision has to be made, the doctor has access only to tiny diagnostic needle biopsies, which often represent less than 0.2% of the prostate volume.
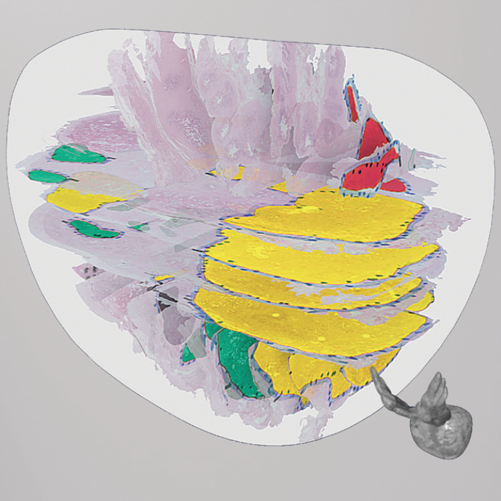
Because prostate cancer tumours are heterogeneous, with biomarkers unevenly distributed throughout the tumour (Figure 2), small needle samples can provide an incomplete picture of the cancer. Tools developed on large surgical specimens therefore are often not directly applicable to biopsy samples, creating a substantial gap between research and clinical benefit.
Improving risk assessment from biopsy samples This project uses deep learning to bridge this gap by adapting a deep learning model (DoMore‑v1‑PCa) previously developed at the Institute for Cancer Genetics and Informatics The model predicts patient outcomes by analysing digital images of standard tissue sections from patients who have undergone surgery. In doing so, it identifies subtle changes in how the tissue looks that are linked to how aggressive the disease is, but are not reflected by the traditional clinical measures. The goal is to further develop and tailor this technology so that it also works when the samples are small needle biopsies.
The AI model is being trained, tested and validated on a unique dataset consisting of nearly 7,000 digitised tissue samples collected from patients treated at the hospital trusts Oslo University Hospital, Vestfold, Vestre Viken and Telemark. Tissue samples from both patients who have undergone surgery and patients monitored under an active surveillance protocol are included.
An innovative approach to clinical decision support The ultimate goal is to integrate the digital AI analyses with traditional clinical measures to create a comprehensive tool for clinical decision-making. This will provide doctors with a safer and more precise basis for distinguishing between patient groups, meaning fewer patients in the uncertain intermediate-risk category. In the longer term, this will help reduce overtreatment, improve outcomes, and contribute to more efficient use of healthcare resources.
The project is funded by the South-Eastern Norway Regional Health Authority (Helse Sør‑Øst) from 2026 to 2029.